-
 South Korea president clings to power after martial law U-turn
South Korea president clings to power after martial law U-turn
-
Presidential vote seen as referendum on Romania's European future

-
 Hamilton bids farewell to Mercedes as Ferrari vie for title
Hamilton bids farewell to Mercedes as Ferrari vie for title
-
New Zealand unchanged in bid to hit back against England

-
 Macron seeks remedy to France's political crisis
Macron seeks remedy to France's political crisis
-
New Natalia Lafourcade album celebrates music's onstage evolutions

-
 Taiwan's Lai kicks off visit to US territory Guam
Taiwan's Lai kicks off visit to US territory Guam
-
Ivory Coast staple cassava meal gains UNESCO heritage status

-
 OpenAI to partner with military defense tech company
OpenAI to partner with military defense tech company
-
Liverpool held but Slot salutes 'special' Salah

-
 Man City needed to break losing 'routine', says Guardiola
Man City needed to break losing 'routine', says Guardiola
-
Leipzig down Frankfurt to reach German Cup quarters, Cologne strike late

-
 Mbappe admits penalty miss 'big mistake' as Bilbao beat Real Madrid
Mbappe admits penalty miss 'big mistake' as Bilbao beat Real Madrid
-
'Sad, disappointed' Mbappe pays penalty as Bilbao beat Real Madrid

-
 US stocks surge to records, shrugging off upheaval in South Korea, France
US stocks surge to records, shrugging off upheaval in South Korea, France
-
Liverpool held in Newcastle thriller, Arsenal inflict Amorim's first defeat

-
 Shiffrin confirms she'll miss Beaver Creek World Cup races
Shiffrin confirms she'll miss Beaver Creek World Cup races
-
Corner kings Arsenal beat Man Utd to close gap on Liverpool

-
 Mbappe pays penalty as Bilbao beat Real Madrid
Mbappe pays penalty as Bilbao beat Real Madrid
-
NFL Jaguars place Lawrence on injured reserve with concussion

-
 North Korea, Russia defence treaty comes into force
North Korea, Russia defence treaty comes into force
-
Openda hits brace as Leipzig beat Frankfurt in German Cup last 16

-
 Schar punishes Kelleher blunder as Newcastle hold Liverpool in thriller
Schar punishes Kelleher blunder as Newcastle hold Liverpool in thriller
-
De Bruyne masterclass helps Man City end seven-game winless streak

-
 Syrian rebels surround Hama 'from three sides', monitor says
Syrian rebels surround Hama 'from three sides', monitor says
-
Lawyers seek leniency for France rape trial defendants, blaming 'wolf' husband

-
 OpenAI chief 'believes' Musk will not abuse government power
OpenAI chief 'believes' Musk will not abuse government power
-
Thousands rally in Georgia after police raid opposition offices

-
 S. Korea opposition push to impeach president
S. Korea opposition push to impeach president
-
Powell 'not concerned' US Fed would lose independence under Trump

-
 French government falls in historic no-confidence vote
French government falls in historic no-confidence vote
-
Syrian White Helmets chief 'dreams' of never pulling a body out of rubble again

-
 NBA Suns lose Durant for at least a week with ankle injury
NBA Suns lose Durant for at least a week with ankle injury
-
Warhammer maker Games Workshop enters London's top stocks index

-
 Iran Nobel winner released for three weeks, 'unconditional' freedom urged
Iran Nobel winner released for three weeks, 'unconditional' freedom urged
-
Red Cross marks record numbers of humanitarians killed in 2024

-
 Johnson's Grand Slam 'no threat', says World Athletics boss Coe
Johnson's Grand Slam 'no threat', says World Athletics boss Coe
-
Qatar's emir and UK's Starmer talk trade as state visit ends

-
 Cuba suffers third nationwide blackout in two months
Cuba suffers third nationwide blackout in two months
-
Russia, Ukraine to send top diplomats to OSCE summit in Malta

-
 Spanish royals to attend memorial service for flood victims
Spanish royals to attend memorial service for flood victims
-
LPGA, USGA new policy requires female at birth or pre-puberty change

-
 Stick to current climate change laws, US tells top UN court
Stick to current climate change laws, US tells top UN court
-
British Museum chief says Marbles deal with Greece 'some distance' away

-
 Pope Francis receives electric popemobile from Mercedes
Pope Francis receives electric popemobile from Mercedes
-
Gaza civil defence: thousands flee Israeli strikes, evacuation calls

-
 Trump names billionaire private astronaut as next NASA chief
Trump names billionaire private astronaut as next NASA chief
-
Pidcock to leave INEOS Grenadiers at end of season

-
 Seoul stocks weaken, Paris advances despite political turmoil
Seoul stocks weaken, Paris advances despite political turmoil
-
South America summit hopes to seal 'historic' trade deal with EU


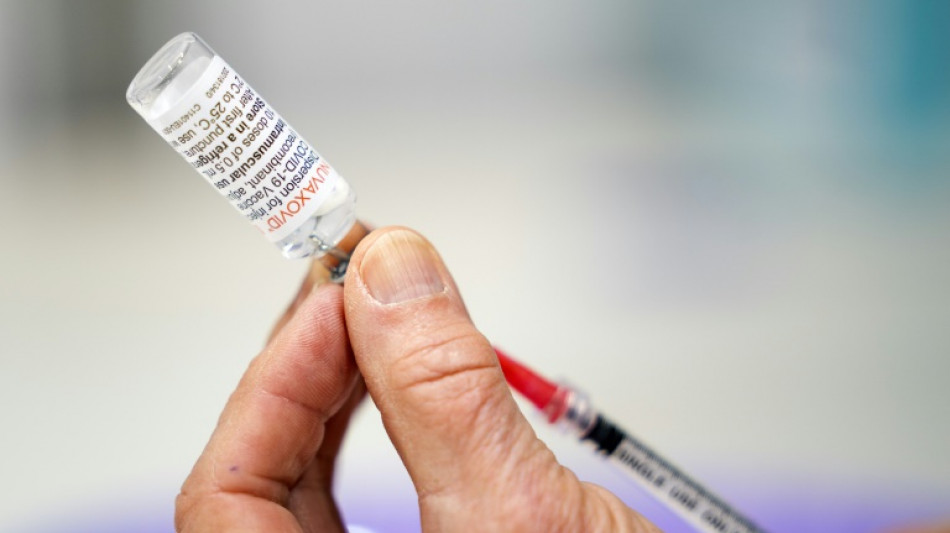
FDA experts weigh authorizing Novavax Covid-19 vaccine in US
A panel of experts convened by the US drug regulator was meeting Tuesday to consider authorizing the Novavax Covid-19 shot, a late runner in the fight against the virus that could nonetheless play a role in overcoming vaccine hesitancy.
Three vaccines are currently approved in the United States: Pfizer and Moderna, which are based on messenger RNA, and Johnson and Johnson, an adenovirus vector vaccine.
But the last of these, the J&J vaccine, was recently restricted in the US after being linked to a rare but serious clotting condition, especially in women of reproductive age.
It is now only recommended for adults who cannot access Pfizer or Moderna for medical or other serious reasons.
The Novavax vaccine was an early frontrunner in the vaccine race, but fell behind after being hit by manufacturing and regulatory delays.
Though the company is American, the US is one of the few major markets where it hasn't yet received authorization -- the EU, UK, Canada, Australia are among many that have already given it the green light.
Officials hope that the shot, which is based on synthetic proteins, could provide an alternative to the mRNA vaccines for people still hesitant.
"We do have a problem with vaccine uptake that is very serious in the United States," Peter Marks, a senior scientist for the Food and Drug administration, said at the start of the meeting.
"And anything we can do to get people more comfortable to be able to accept these potentially life saving medical products, is something that we feel we are compelled to do."
Of the various vaccine technologies, mRNA has been subject to the most misinformation efforts.
Novavax's vaccine was found to be 90 percent effective against symptomatic cases of the disease, in trials conducted before the appearance of the Omicron variant, according to the FDA.
But six cases of myocarditis, an inflammation of the heart muscle, were detected in the group that received the vaccine, against one case in the placebo group, in a trial of around 40,000 people.
Novavax says there is insufficient evidence to establish a causal relationship between the cases of myocarditis and the vaccine.
The FDA voiced concern over the myocarditis link on Friday, sending Novavax shares to drop 20 percent on the New York Stock Exchange. And trading of Novavax stock was halted on Monday pending news from the FDA panel.
Known as a protein subunit vaccine, Novavax is administered in two doses.
It uses a synthetic version of the virus' spike protein to evoke an immune response.
The same technique is used in vaccines against whooping cough, meningococcal meningitis and hepatitis B.
M.Furrer--BTB




